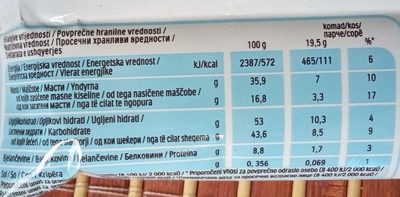
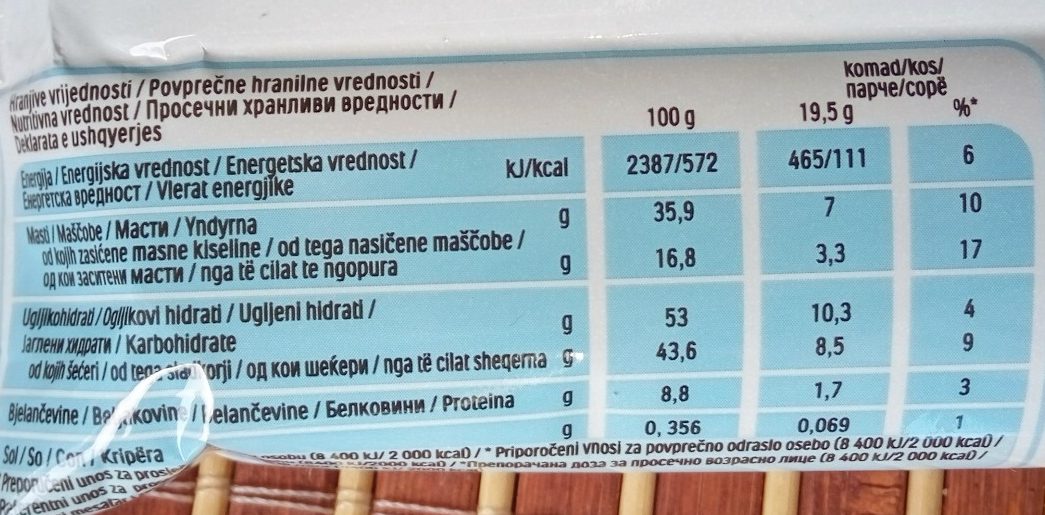
Kinder Bueno White - 39 g
Some of the data for this product has been provided directly by the manufacturer FERRERO FRANCE COMMERCIALE.
Barkod: 80761761
Common name: Vafel proizvod prekriven belom čokoladom sa punjenjem od mleka i lešnika
Količina: 39 g
Ambalaža: en:Plastic, PP 5 - полипропилен, en:mixed plastic film-packet
Брендови: Kinder, Mixte, Kinder bueno
Категорије: en:Snacks, en:Sweet snacks, en:Cocoa and its products, en:Confectioneries, en:Bars, en:Biscuits and cakes, en:Chocolate candies, Бисквит, en:Cereal bars, en:bars-covered-with-chocolate, en:Chocolate cereal bars, en:Chocolate biscuity bars, en:Chocolate confectionery with dairy filling, en:Десерт
Oznake, sertifikati, nagrade:
Tidy man, Triman
Poreklo sastojaka: en:Unspecified
Mesto u kojem se proizvodi ili prerađuje: Poland
Link to the product page on the official site of the producer: http://www.ferrero.fr/fc-2738/#nav=kinde...
Prodavnice: Lidl, Kaufland, Leclerc, Carrefour, Auchan
Државе у којима се продаје: Белгија, Бугарска, Француска, Немачка, Мађарска, Италија, Пољска, Румунија, Србија, Швајцарска
Matching with your preferences
Other information
Conservation conditions: A conserver au sec et à l'abri de la chaleur
Customer service: CS90058 76136 MONT-SAINT-AIGNAN
Report a problem
Data sources
The manufacturer FERRERO FRANCE COMMERCIALE uses Equadis to automatically transmit data and photos for its products.
Product added on од openfoodfacts-contributors
Last edit of product page on од foodless.
Страницу производа такође уредио aleene, annelotte, arc2, asmoth, ayyyvocado, bojackhorseman, bugmenot, cautery-mild0u, choosebetter, date-limite-app, davidepio, desan, feat, ferrero, foodrepo, foodvisor, g123k, guilvareux, inf, insectproductadd, jeanbono, julien000, kiliweb, musarana, nicolasleger, nnejma, october-food-facts, org-ferrero-france-commerciale, packbot, pyrka, roboto-app, scanbot, tacite, tatas, telperion87, thaialagata, yuka.R0tBZFA0RXgrZGNBZ2MxZzkwM081STVTNWJxblIzcXVNdGdzSVE9PQ, yuka.R3A0UUU0VWxoT0FtbC9SdW9SM255czliNkorSkJYS2NGK1ZCSUE9PQ, yuka.RzZJQkRwWUNtOVFrbThNMzN4UHhvZlYzbVpTRFpFSHRNTnNYSWc9PQ, yuka.UTVrYklwd3UvTUVqdC9GazRrbmVvL1Vxd2FPcFduaXdDODlJSVE9PQ, yuka.UXJ4WUZxQTlqZEk3dU1JM3dnTGwzUDk2bUwySGVscTNHY3doSVE9PQ, yuka.UjV3d0hKUWpsdm9OdnN3YW95M2JvZHg3MkxtcGZFcXZFZTRJSWc9PQ, yuka.UlA4dlNyZzh0dUFWeGZCa3BDak53dU44NFlYM1VqS3NKYkJQSVE9PQ, yuka.V28wcERmVWduOTVYdlBBRDRSUGIyTlpjNG9PUUJWanBHKzQrSVE9PQ, yuka.VFk4SERLUVkrT2NLd05vaTRDN28rZlZ2L1lDd1pVam9JYklXSVE9PQ, yuka.VGFZa0xyVXQvOTREeHNRUjJFbm4xUHhreUxHd1ZrU2VPZlkvSVE9PQ, yuka.VjZrZlM0QWFscVFJc2RnWi95L3p4YzEvNXBDT0RWR2FKdlpQSVE9PQ, yuka.VnFvN0FJY2cvUDhoeFBkdTVUYnIxTjFPN0s2SVkxL3RETE11SVE9PQ, yuka.WFpFK0NKVW9pK0F2Z2NjbDNoRCsvZmxxMzgrd1FVeXlMdU10SVE9PQ, yuka.ZElVSFR2UVlucVZidHRnZXpqbnEyL1phMklhMVoybU5EK3BOSVE9PQ, yuka.ZmYwL0Nhc1JwY1pRbS9jQjJqSG44UDF4eTYrVlltZVNOTmdQSWc9PQ, yuka.sY2b0xO6T85zoF3NwEKvlk5ECdXM8jjlCD3viRSa_PmQHMf2Z95t25LZHas, yuka.sY2b0xO6T85zoF3NwEKvllZFUYLUhiqbGQbhoWeC4vCKcMP0W_dO5YWlFqs, yukafix, zdrowa-polska, zi, zixianglim.